Foamix Pharmaceuticals Ltd. issued the following announcement on Oct. 17.
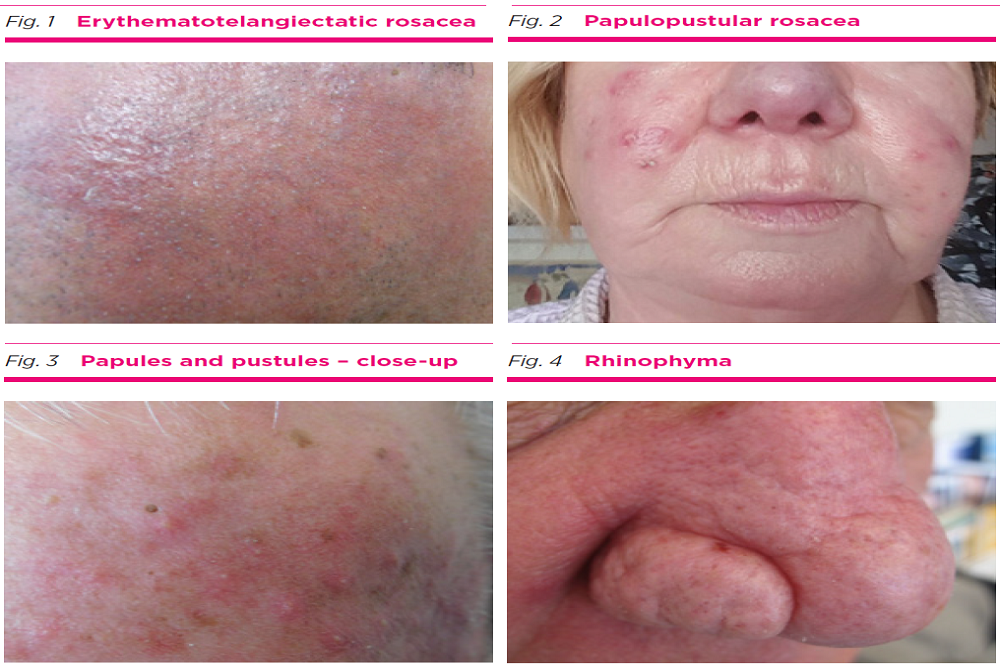
Foamix Pharmaceuticals Ltd. (Nasdaq: FOMX), a clinical stage specialty pharmaceutical company focused on developing and commercializing proprietary topical therapies to address unmet needs in dermatology, today announced that the U.S. Food and Drug Administration (FDA) has accepted for review the New Drug Application (NDA) for FMX103 (minocycline topical foam 1.5%) topical foam. Foamix is seeking approval of FMX103 for the treatment of moderate-to-severe papulopustular rosacea in adults. The FDA has set June 2nd, 2020 as the Prescription Drug User Fee Act (PDUFA) action date.
"The FDA's acceptance of the FMX103 NDA is another important milestone for Foamix as the company evolves into a fully integrated pharmaceutical company with clinical, development, and commercial capabilities. We look forward to working closely with the FDA throughout the review process," said David Domzalski, Chief Executive Officer. "The application includes what we believe is a strong and complete clinical data package. Rosacea is a challenging condition to treat for patients and healthcare providers. If approved, FMX103 has the potential to address significant unmet needs for those who are burdened with rosacea."
The NDA submission is supported by the previously communicated results from two 12-week double-blind Phase 3 efficacy and safety trials (Studies FX2016-11 and FX2016-12) and one 40-week open-label safety extension trial (Study FX2016-13). In Studies FX2016-11 and FX2016-12, FMX103 met both co-primary endpoints, demonstrating statistically significant improvements in inflammatory lesion count and Investigator Global Assessment (IGA) treatment success. No treatment-related serious adverse events have been identified in the FMX103 clinical development program, where the most common adverse event was upper respiratory tract infection. The NDA submission also incorporates information from Phase 1 and Phase 2 clinical trials, chemistry manufacturing and controls, and data from nonclinical toxicology studies.
About Foamix Pharmaceuticals
Foamix is a late clinical-stage specialty pharmaceutical company working to solve some of today's most difficult therapeutic challenges in dermatology and beyond.
With expertise in topical medicine innovation as a springboard, the Company is working to develop and commercialize solutions that were long thought impossible, including the world's first topical minocycline foam. Its proprietary Molecule Stabilizing Technology (MST™) is utilized in the Company's dermatology products in late stage development: FMX101 for the potential treatment of inflammatory lesions of non-nodular moderate to severe acne vulgaris in adults and pediatric patients 9 years of age and older, and FMX103 for the potential treatment of moderate-to-severe papulopustular rosacea in adults.
Foamix is a different type of specialty pharmaceutical company by design, driven to see the solutions, overcome barriers in all aspects of business, and reimagine what's possible for conditions with high unmet needs.
Original source can be found here.




