Latest News
U.S. FOOD AND DRUG ADMINISTRATION: FDA expands approval of treatment for cystic fibrosis to include patients ages 6 and older
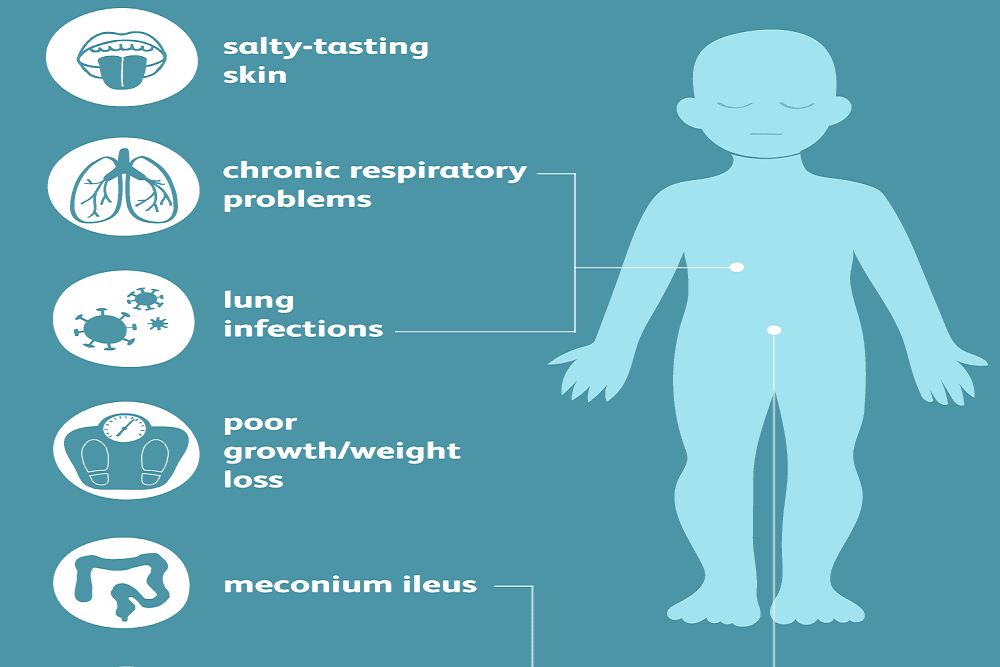
The U.S. Food and Drug Administration expanded the indication for Symdeko (a combination of tezacaftor/ivacaftor) tablets for treatment of pediatric patients ages 6 years and older with cystic fibrosis who have certain genetic mutations.
U.S. FOOD AND DRUG ADMINISTRATION: FDA approves new treatment for hypoactive sexual desire disorder in premenopausal women
The U.S. Food and Drug Administration approved Vyleesi (bremelanotide) to treat acquired, generalized hypoactive sexual desire disorder (HSDD) in premenopausal women.
FDA launched 7 foodborne disease probes in first 6 months of 2019
Seven outbreaks of foodborne illnesses have been reported so far this year, according to the U.S. Food and Drug Administration.
JOHNSON & JOHNSON: Janssen Announces U.S. FDA Approval of DARZALEX® (daratumumab) in Combination with Lenalidomide and Dexamethasone for Newly Diagnosed Patients with Multiple Myeloma Who Are Transplant Ineligible
Combination regimen reduced the risk of disease progression or death by 44 percent in newly diagnosed patients who are transplant ineligible
New non-insulin treatment for type 2 diabetes approved
The Food and Drug Administration recently approved the first non-insulin drug approved to treat type 2 diabetes in pediatric patients since metformin was approved for pediatric use in 2000.
Initial FDA tests find little fluoroalkyl compounds in foods
The FDA has been working on methods to determine how much per- and polyfluoroalkyl substances (PFAS) are in foods. Initial testing was done on foods common in the American diet.
Life Rising’s dietary supplements seized over potential consumer risk
U.S. Marshals seized over 300,000 containers of Life Rising Corporation’s dietary supplements in a raid requested by the Food and Drug Administration.
U.S. FOOD AND DRUG ADMINISTRATION: Statement on stem cell clinic permanent injunction and FDA’s ongoing efforts to protect patients from risks of unapproved products
U.S. District Judge Ursula Ungaro of the Southern District of Florida issued an order that stops US Stem Cell Clinic LLC, of Weston, Florida, and US Stem Cell Inc., of Sunrise, Florida, and their Chief Scientific Officer Kristin Comella, Ph.D., from manufacturing or distributing any and all stromal vascular fraction (SVF) products, which are adipose (fat) tissue derived stem cell products, until they come into compliance with the law.
U.S. FOOD AND DRUG ADMINISTRATION: FDA approves first treatment for neuromyelitis optica spectrum disorder, a rare autoimmune disease of the central nervous system
The U.S. Food and Drug Administration approved Soliris (eculizumab) injection for intravenous use for the treatment of neuromyelitis optica spectrum disorder (NMOSD) in adult patients who are anti-aquaporin-4 (AQP4) antibody positive. NMOSD is an autoimmune disease of the central nervous system that mainly affects the optic nerves and spinal cord.
FDA warns of infection risk from fecal microbiota transplants
Patients who receive fecal microbiota for transplant (FMT) are at risk of life-threatening infections, the Food and Drug Administration said.
U.S. FOOD AND DRUG ADMINISTRATION: Statement on steps to make health care professional and patient labeling information for prescription medications consistent and clear
The FDA is committed to making sure that health care professionals and patients have up-to-date, accurate and actionable information about prescription medications so that informed treatment decisions can be made. We recognize that the choice of which medication to use, if any, for a particular patient is often a complex decision.
U.S. FOOD AND DRUG ADMINISTRATION: FDA issues third status report on investigation into potential connection between certain diets and cases of canine heart disease
The U.S. Food and Drug Administration is providing an update on its investigation into reports of dilated cardiomyopathy (DCM) in dogs eating certain types of pet food.
PUMA BIOTECHNOLOGY: Submits a Supplemental New Drug Application to U.S. FDA for Neratinib to Treat HER2-Positive Metastatic Breast Cancer
Puma Biotechnology, Inc. (NASDAQ: PBYI), a biopharmaceutical company, has submitted a supplemental New Drug Application (sNDA) to the U.S. Food and Drug Administration (FDA) for neratinib in combination with capecitabine for the treatment of patients with HER2-positive metastatic breast cancer who have failed two or more prior lines of HER2-directed treatments (third-line disease).
PERRIGO COMPANY PLC: Perrigo Announces FDA Final Approval and Launch Of The AB-rated Generic Version Of Metrogel-Vaginal® Gel
Perrigo Company plc (NYSE; TASE: PRGO) announced its product development partner received final approval from the U.S. Food and Drug Administration for its AB-rated Abbreviated New Drug Application ("ANDA") referencing Metrogel-Vaginal® (metronidazole vaginal gel 0.75%).
U.S. FOOD AND DRUG ADMINISTRATION: FDA approves new treatment for refractory multiple myeloma
The U.S. Food and Drug Administration granted accelerated approval to Xpovio (selinexor) tablets in combination with the corticosteroid dexamethasone for the treatment of adult patients with relapsed refractory multiple myeloma (RRMM) who have received at least four prior therapies and whose disease is resistant to several other forms of treatment, including at least two proteasome inhibitors, at least two immunomodulatory agents, and an anti-CD38 monoclonal antibody.
KARYOPHARM THERAPEUTICS: Karyopharm Announces FDA Approval of XPOVIO™ (selinexor) for the Treatment of Patients with Relapsed or Refractory Multiple Myeloma
XPOVIO is the First and Only Nuclear Export Inhibitor Approved by the FDA
6 drugs recalled during June, FDA reports
Six drugs were recalled in June to protect patients from possible harmful defects associated with the products, the U.S. Food and Drug Administration reported.
REDHILL BIOPHARMA: Announces FDA Acceptance of New Drug Application for Talicia®
U.S. FDA accepts NDA for priority review
INSYS THERAPEUTICS, INC: FDA Accepts New Drug Application for INSYS Therapeutics’ Naloxone Nasal Spray for the Emergency Treatment of Known or Suspected Opioid Overdose
INSYS Therapeutics, Inc. (OTCMKTS: INSYQ), announced that its New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) for its investigational, proprietary naloxone nasal spray for the emergency treatment of known or suspected opioid overdose in both adult and pediatric patients has been accepted for filing.