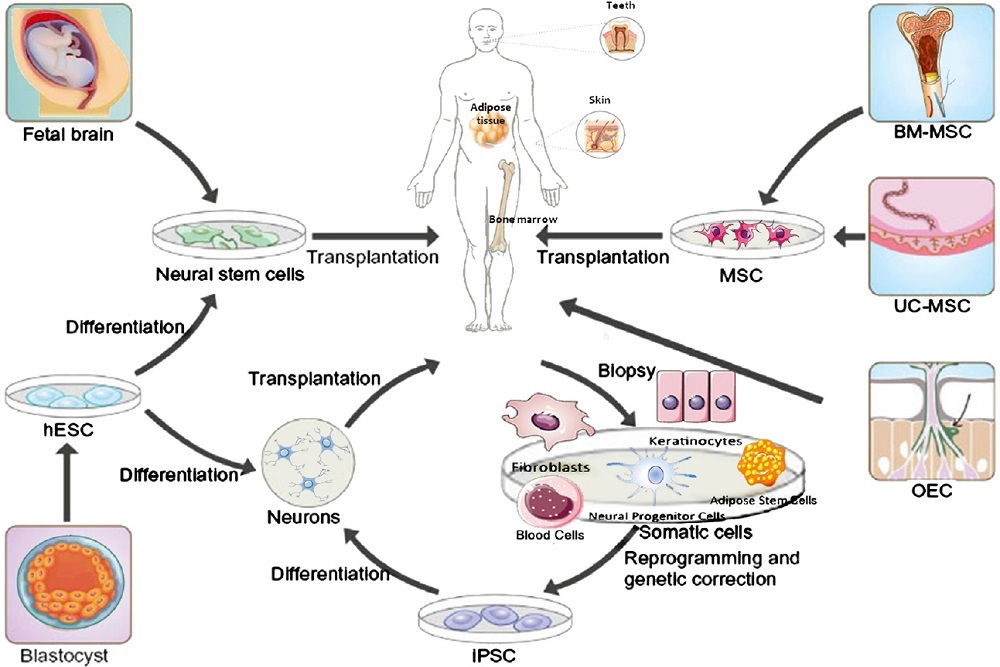
ALLERGAN PLC: Allergan and Molecular Partners Announce Acceptance of U.S. FDA Biologics License Application and Validation of EMA Marketing Authorisation for Abicipar pegol in Patients with Neovascular (Wet) Age-related Macular Degeneration
Filing includes data from two Phase 3 trials which evaluated the safety and efficacy of Abicipar quarterly dosing regimen